Colchicine for the treatment of COVID-19 patients: efficacy, safety, and model informed dosage regimens
- PMID: 33845715
- PMCID: PMC8054498
- DOI: 10.1080/00498254.2021.1909782
Colchicine for the treatment of COVID-19 patients: efficacy, safety, and model informed dosage regimens
Abstract
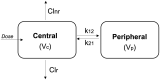
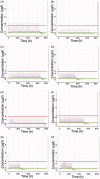
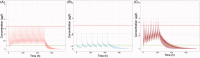
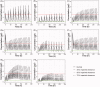
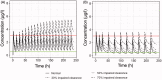
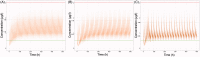
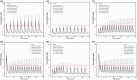
Colchicine is widely investigated for cardioprotection of COVID-19 patients since it can prevent the phenomenon of 'cytokine storm' and may reduce the complications arising from COVID-19. Despite the potentially beneficial effects of colchicine, there is no consensus on the appropriate dosage regimen and numerous schemes are currently used.In this study, simulations were performed to identify the ability of dosage regimens to attain plasma levels in CVOID-19 patients, known to be generally safe and efficacious. Since renal and hepatic impairment, as well as, drug-drug interactions have been identified to be the most significant factors increasing colchicine toxicity, the impact of these interactions was assessed in the simulations.Some dosage regimens lead to high colchicine concentrations, while others result in sub-therapeutic levels. Additional dosage schemes were proposed in this study aiming to be applied in patients with clearance insufficiency. Colchicine administration of 0.5 mg twice daily, can be considered safe and effective. In cases of clearance impairment, doses as low as 0.25 mg thrice or twice daily should be applied.Colchicine is a narrow therapeutic index drug and dosage regimens tailored to patients' needs should be designed.
Keywords: COVID-19; Colchicine; cardioprotection; dosage regimens; pharmacokinetics; simulations.
Conflict of interest statement
The authors report no declarations of interest.
Figures
Similar articles
-
Colchicine and SARS-CoV-2: Management of the hyperinflammatory state.Respir Med. 2021 Mar;178:106322. doi: 10.1016/j.rmed.2021.106322. Epub 2021 Feb 1. Respir Med. 2021. PMID: 33550151 Free PMC article. Review.
-
Proactive anti-inflammatory therapy with colchicine in the treatment of advanced stages of new coronavirus infection. The first results of the COLORIT study.Kardiologiia. 2021 Mar 1;61(2):15-27. doi: 10.18087/cardio.2021.2.n1560. Kardiologiia. 2021. PMID: 33734043 Clinical Trial.
-
Sequential doxycycline and colchicine combination therapy in Covid-19: The salutary effects.Pulm Pharmacol Ther. 2021 Apr;67:102008. doi: 10.1016/j.pupt.2021.102008. Epub 2021 Mar 14. Pulm Pharmacol Ther. 2021. PMID: 33727066 Free PMC article.
-
PROTECT Trial: A cluster-randomized study with hydroxychloroquine versus observational support for prevention or early-phase treatment of Coronavirus disease (COVID-19): A structured summary of a study protocol for a randomized controlled trial.Trials. 2020 Jul 31;21(1):689. doi: 10.1186/s13063-020-04527-4. Trials. 2020. PMID: 32736597 Free PMC article.
-
Lopinavir/ritonavir: a review of its use in the management of HIV infection.Drugs. 2003;63(8):769-802. doi: 10.2165/00003495-200363080-00004. Drugs. 2003. PMID: 12662125 Review.
Cited by 5 articles
-
Severe post-COVID-19 costochondritis in children.Proc (Bayl Univ Med Cent). 2021 Sep 27;35(1):56-57. doi: 10.1080/08998280.2021.1973274. eCollection 2022. Proc (Bayl Univ Med Cent). 2021. PMID: 34966216 Free PMC article.
-
Immunologic Dysregulation and Hypercoagulability as a Pathophysiologic Background in COVID-19 Infection and the Immunomodulating Role of Colchicine.J Clin Med. 2021 Oct 31;10(21):5128. doi: 10.3390/jcm10215128. J Clin Med. 2021. PMID: 34768648 Free PMC article. Review.
-
Colchicine Is Safe Though Ineffective in the Treatment of Severe COVID-19: a Randomized Clinical Trial (COLCHIVID).J Gen Intern Med. 2021 Nov 9:1-11. doi: 10.1007/s11606-021-07203-8. Online ahead of print. J Gen Intern Med. 2021. PMID: 34755269 Free PMC article.
-
Drug-Induced Liver Injury in COVID-19 Patients: A Systematic Review.Front Med (Lausanne). 2021 Sep 20;8:731436. doi: 10.3389/fmed.2021.731436. eCollection 2021. Front Med (Lausanne). 2021. PMID: 34616757 Free PMC article.
-
Consensus Statement Regarding the Efficacy and Safety of Long-Term Low-Dose Colchicine in Gout and Cardiovascular Disease.Am J Med. 2022 Jan;135(1):32-38. doi: 10.1016/j.amjmed.2021.07.025. Epub 2021 Aug 18. Am J Med. 2022. PMID: 34416165 Free PMC article. Review.
References
-
- Achtert, G., Scherrmann, J.M., and Christen, M.O., 1989. Pharmacokinetics/bioavailability of colchicine in healthy male volunteers. European journal of drug metabolism and pharmacokinetics, 14 (4), 317–322. - PubMed
-
- Audemard-Verger, A., et al. , 2017. Characteristics and management of IgA vasculitis (Henoch-Schönlein) in adults: data from 260 patients included in a French multicenter retrospective survey. Arthritis & rheumatology, 69 (9), 1862–1870. - PubMed
-
- Ben-Chetrit, E. and Levy, M., 1998. Colchicine: 1998 update. Seminars in arthritis and rheumatism, 28 (1), 48–59. - PubMed
-
- Berkun, Y., et al. , 2012. Pharmacokinetics of colchicine in pediatric and adult patients with familial Mediterranean fever. International journal of immunopathology and pharmacology, 25 (4), 1121–1130. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
